![The Synthesis of Methyl-Substituted Spirocyclic Piperidine-Azetidine (2,7-Diazaspiro[3.5]nonane) and Spirocyclic Piperidine-Pyrrolidine (2,8-Diazaspiro[4.5]decane) Ring Systems | The Journal of Organic Chemistry The Synthesis of Methyl-Substituted Spirocyclic Piperidine-Azetidine (2,7-Diazaspiro[3.5]nonane) and Spirocyclic Piperidine-Pyrrolidine (2,8-Diazaspiro[4.5]decane) Ring Systems | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.5b02890/asset/images/medium/jo-2015-028905_0009.gif)
The Synthesis of Methyl-Substituted Spirocyclic Piperidine-Azetidine (2,7-Diazaspiro[3.5]nonane) and Spirocyclic Piperidine-Pyrrolidine (2,8-Diazaspiro[4.5]decane) Ring Systems | The Journal of Organic Chemistry
Welcome to Chem Zipper.com......: Pyridine is almost 1 million times less basic than piperidine? Why?

Molecular Modeling of Local Anesthetic Drug Binding by Voltage-Gated Sodium Channels | Molecular Pharmacology

Recent Advances in the Total Synthesis of Piperidine and Pyrrolidine Natural Alkaloids with Ring‐Closing Metathesis as a Key Step - Felpin - 2003 - European Journal of Organic Chemistry - Wiley Online Library

Lewis Acid-Catalyzed Ring-Opening Reactions of Semicyclic N,O-Acetals Possessing an Exocyclic Nitrogen Atom: Mechanistic Aspect and Application to Piperidine Alkaloid Synthesis | Journal of the American Chemical Society
Piperidine nucleus in the field of drug discovery | Future Journal of Pharmaceutical Sciences | Full Text

Structures of some representative natural piperidine alkaloids, from... | Download Scientific Diagram
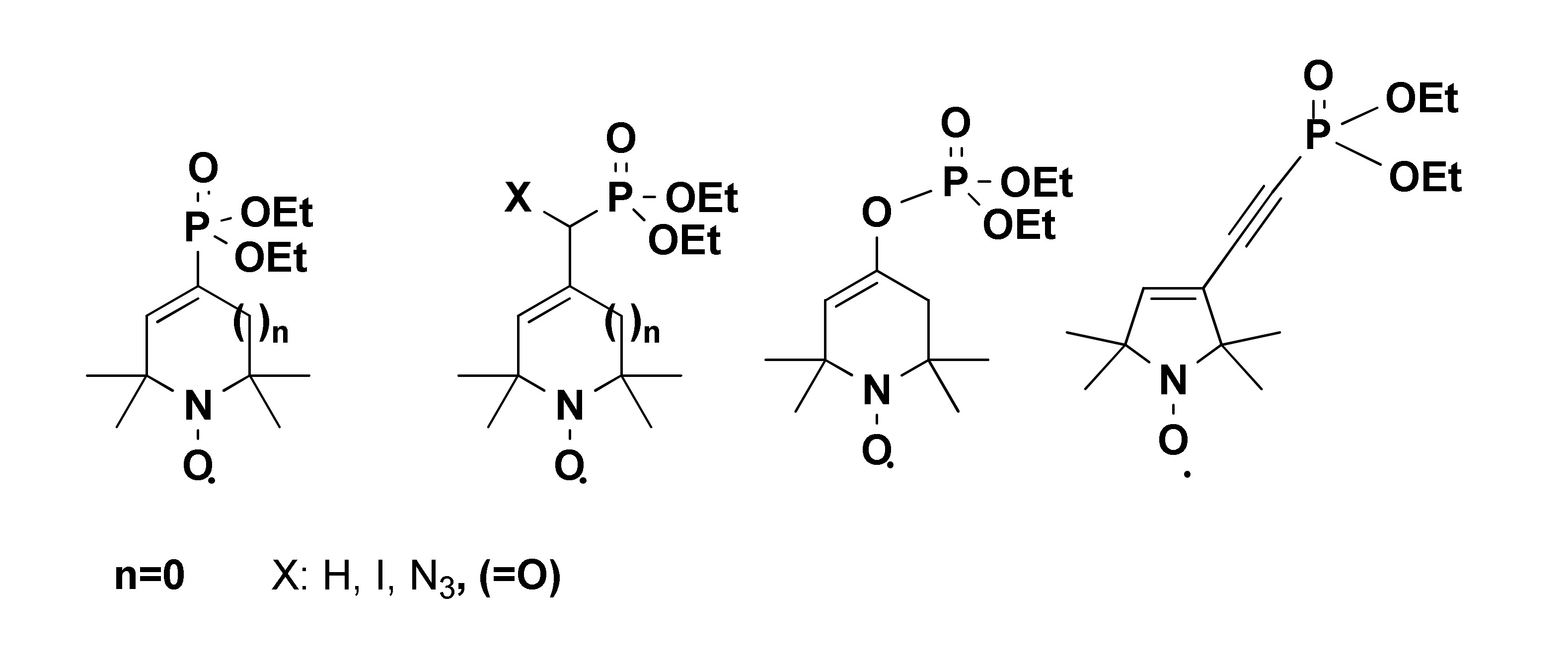
Molecules | Free Full-Text | Syntheses and Reactions of Pyrroline, Piperidine Nitroxide Phosphonates

Azetidiniums: Ring‐Expansion to Pyrrolidines, Piperidines, Azepanes, and Azocanes - Masson - 2020 - European Journal of Organic Chemistry - Wiley Online Library
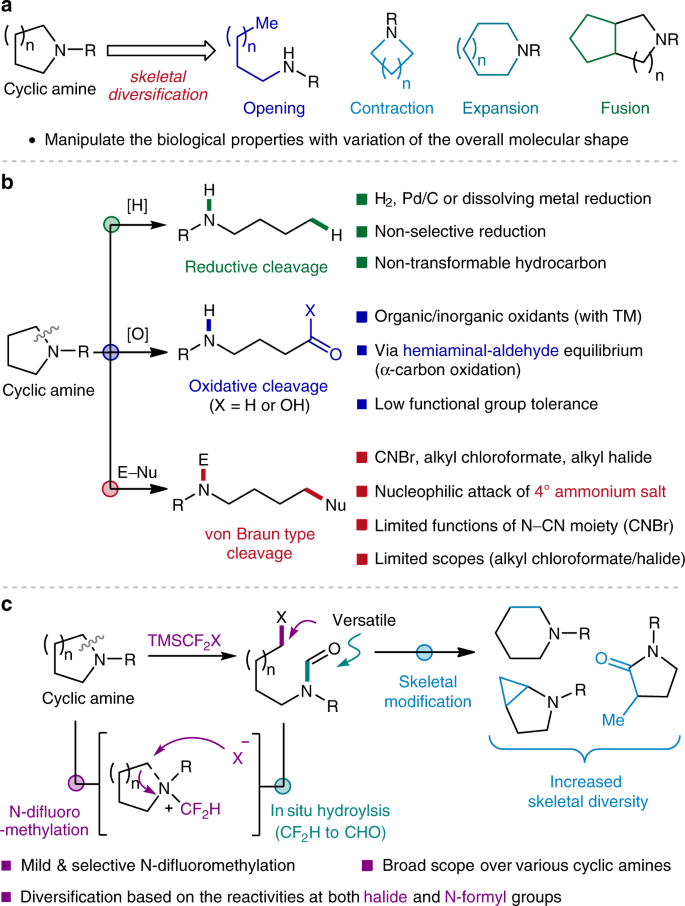
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications


![PDF] Use of Piperidine and Pyrrolidine in Knoevenagel Condensation | Semantic Scholar PDF] Use of Piperidine and Pyrrolidine in Knoevenagel Condensation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/204d8cfb50b6267ae364d6e6a5a3077f018f4e9a/2-Figure2-1.png)